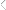The uses are endless…
•;Transferring;to;or;from;in-house;to;shipping;pallets;or;slip;sheets
•;Replacing;damaged;items;at;the;bottom;of;a;load
•;Turning;inventory;for;freshness
•;Replacing;broken;pallets
For complete details, visit www.SouthworthProducts.com/inverter
TEL: (800) 743-1000 • FAX: (207) 797-4734
SouthworthProducts.com;•;salesinfo@SouthworthProducts.com
14-405 Pallet Invertor Ad-DCV.indd 1 3/3/15 9: 23 AM
chain rules, replacing the patchwork of
state regulations that preceded the federal standard, according to Eric Newmark,
program director for industry cloud and
commercial life sciences at IDC Health
Insights.
But the major benefits won’t arrive until
2023, when the FDA fully implements
its item-level and package-level visibility
requirements. This will foster an efficient
product recall process and enhance law
enforcement efforts, experts said.
“There are enormous inefficiencies …
throughout the industry; this is a problem
in the billions of dollars per year in the
global industry,” Newmark said. “This is
a huge guessing game. If you don’t have
visibility, you have to recall 10 times as
many drugs.”
CHALLENGES FOR SHIPPING DRUGS
When healthcare companies choose a 3PL
to distribute their products, they should
make sure the partner can overcome some
common hurdles.
One problem with the precision tracking
requirements is that warehouse workers
can’t retract a published ASN to correct a
simple loading mistake. If workers ship 11
crates instead of 10, the transaction histories won’t match up and the shipment will
have to sit idle until the corrected paperwork catches up. That can be a slow and
costly process.
Another common problem is shipping
a load to the proper customer but to
the wrong location. Some pharmaceutical
products are temperature controlled, and
the receiver may not want to return a valuable product and risk its becoming stale in
warm weather. That can result in another
costly delay while replacements are sent.
A third challenge may be training a
warehouse management system (WMS) to
recognize exceptions to DSCSA-controlled
products. Most WMS platforms have no
trouble tracking a batch of prescription
pills, but the same pharmaceutical manufacturer may also ship related materials
that do not fall under the act, such as over-the-counter medicines and simple medical
devices that do not require strict tracking.
SOLUTION MAY BE IN THE CLOUD
Distribution center managers may not have
to buy new material handling or
tracking systems to comply with
the pharmaceutical tracking law,
but they will probably need to train
floor workers and logistics coor-
dinators to manage the immense
amount of data that will be created
under the new regulatory regime.
“Visualization of products stops
now at the lot level, but when you
get down to the case and pallet
level, and then the item level, you
will be looking at a thousand or
ten thousand times as much data
being created, so there will be an
enormous influx of data,” said
Newmark of IDC. “And they will
need the ability to handle, organize,
sort, and store all that data. That
will be interesting.”
That is why many business-
es are choosing to bypass on-site
servers entirely and subscribe to
cloud-based supply chain transac-
tion management solutions from
providers like Axway, rfXcel Corp.,
and SAP.
“When the FDA comes calling,
you’d better have maintained those
documents,” said Shabbir Dahod,
CEO of North Reading, Mass.-
based TraceLink, which offers a
cloud-based track and trace net-
work to subscribers. “As you pick
and pack from your DC, you have
to correlate that with the transac-
tion history to prove the link to the
product or … that it was shipped to
customers A to Z.”
Smaller companies may not have
the budget for these requirements,
and even larger players may choose
to avoid incurring the extra cost of
IT support. By using a cloud-based
solution, a 3PL provider can use
a Web browser to track and trace
all of its products, manage large-scale serialization of drug units, and
exchange data with its trading partners, experts said.